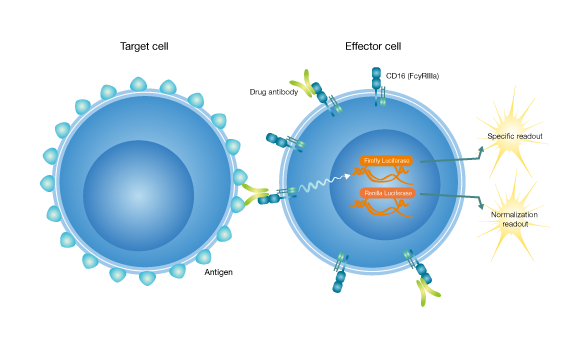
Ideal assays for determining the specificity or unwanted activity of ADCC inducing compounds
A recent successful range of therapeutic antibodies depend on effector functions for their mode-of-action. Here the constant fc-regions of the antibody interact with fc-receptors on the surfaces of immune cells.
Such interactions can result in the killing of a target cell, e.g., a virus-infected cell, through immune effector processes like antibody-dependent cell-mediated cytotoxicity (ADCC) and antibody-dependent cellular phagocytosis (ADCP). In addition, the fc-region can bind to the complement component C1q and cause complement-dependent cytotoxicity (CDC).
While these effects are something that these new therapeutics is harnessing in the treatment of different disease, in particular different cancer forms, the existence of such effects must be thoroughly investigated to ensure the safety of the drug and to avoid adverse effects.