- Other Products
- CCP
In antibody-dependent cell-mediated cytotoxicity (ADCC), Fc receptor-bearing effector cells recognize and kill antibody-coated target cells that express tumor or pathogen-specific antigens on their surface.
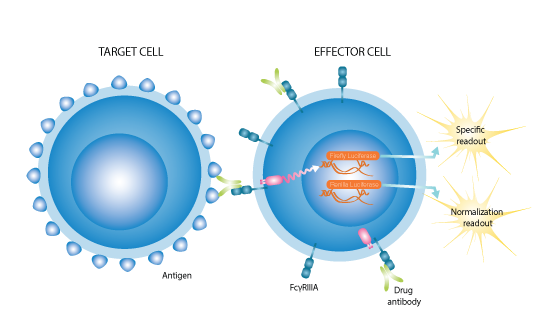

In antibody-dependent cell-mediated cytotoxicity (ADCC), Fc receptor-bearing effector cells recognize and kill antibody-coated target cells that express tumor or pathogen-specific antigens on their surface.
The term “target cell” indicates a cell in the body that has been infected by a virus or damaged in some way, triggering the production of special proteins that are not found in the body under normal circumstances. Antigens on the surface of the cell act as red flags that cause the immune system to attack and destroy the target cells.
Contrary to what happens in the human body, the effector cells of ADCC reporter gene assays do not kill the target cells. Instead, a signaling pathway is activated in the effector cell upon binding to the Fc-region of a target-cell bound antibody that results in the expression of luciferase. The amount of luciferase generated correlates to the ability of the system to elicit ADCC.
The target cells are important in this mechanism since it requires a crosslink, with an antibody between the target cells and the effector cell, in order to get a signal/action.
The target cells are essential in this mechanism as a crosslink between the target cell, antibody and effector cell is required to get a signal.
As described above, a critical feature of activating ADCC is the antigen-presenting target cell. Traditionally, wild-type cells that naturally express this antigen have been used in ADCC bioassays. However, this strategy is not without problems, as antigen levels on the cell surface can vary based on passage and when the cells were harvested, which can make it difficult to get clear and reproducible results.
A novel way of creating target cells is to use engineered cells that overexpress the antigen, resulting in a high constant level of the antigen at the cell surface, which results in a sensitive and reproducible assay. Another benefit of this technique is the availability of control cell lines where the gene encoding the antigen has been made non-functional using CRISPR/Cas 9 editing.
Engineered target cells and their control cells can be used to determine differences in ADCC activity with high precision and specificity. In addition, the antigen negative control target cells provide a means for detecting and correcting nonspecific effects sometimes observed in human serum samples.
The ADCC activity mediated by a therapeutic antibody can be examined in more detail when engineered target cells with different level of expression/deletional mutations in expression can be assesses side by side and compared. These results can indicate if an ADCC‐dependent antitumor activity results from affinity of therapeutic mAb to the extracellular domain of the antigen and/or if this activity is independent of mutation status of the antigen.
Engineered target cells offer excellence performance when determining ADCC activity mediated by therapeutic antibodies. Side-by-side comparisons of engineered target cells that express varying levels of antigen, allow researchers to analyze ADCC activity in great detail. These results can indicate if an ADCC‐dependent antitumor activity results from the affinity of the therapeutic mAb to the extracellular domain of the antigen or if the activity is independent of the mutation status of the antigen.
Combined use of cell-based reporter bioassays with target cells engineered to express membrane-bound soluble ligands can provide a simple, specific, and quantitative platform to measure Fc-mediated effector functions of therapeutic antibodies. It can also improve accuracy and routine GMP performance.
Overexpression of the antigen on the surface of the target cells can give more consistent ADCC activity and higher signal, which means a larger dynamic range for the assays.
Lallemand and colleagues found that overexpressing the CD20 gene resulted in a constant high level of CD20 at the surface of CD20+ target cells. This level was approximately 16-fold higher than wild-type Raji cells and resulted in significantly higher ADCC activity (Lallemand et al. J. Immunol. Res. 2017: 3908289).