- Other Products
- CCP
In the late 1800s, William B. Coley used crude bacterial extracts to treat tumor patients. He found that the extracts were effective in inducing tumor necrosis. However, patients treated with his extracts were also found to suffer from a severe systematic inflammatory reaction. One of the major factors in stimulating such inflammatory reactions in humans is now known as tumor necrosis factor alpha (TNFα).

In the late 1800s, William B. Coley used crude bacterial extracts to treat tumor patients. He found that the extracts were effective in inducing tumor necrosis. However, patients treated with his extracts were also found to suffer from a severe systematic inflammatory reaction. One of the major factors in stimulating such inflammatory reactions in humans is now known as tumor necrosis factor alpha (TNFα).
TNFα plays many important physiological roles in humans. It’s involved in tumor cell necrosis and apoptosis, but also other processes such as embryo development, sleep-wake cycle regulation, fever and in the defense against viral and bacterial infections. In addition, TNFα is an important player in the systematic inflammatory reactions, not only by its own secretion but also by stimulating other inflammatory cytokines and chemokines.
A high dose of regional TNFα causes necrosis and destruction of tumor blood vessels, as well as enhances the effect of chemotherapy. This strategy has been used for treatment of metastatic melanoma and soft tissue sarcoma.
Interestingly, TNFα is also involved in the pathogenesis of cancer-associated chronic inflammation and in autoimmune disorders, which has led to the development of anti-TNFα drugs. Several antibody-based therapies have been approved and several more are in the pipeline. They have been very effective in treating autoimmune disorders such as rheumatoid arthritis, but have had side-effects that include an increased risk of developing several forms of cancer.
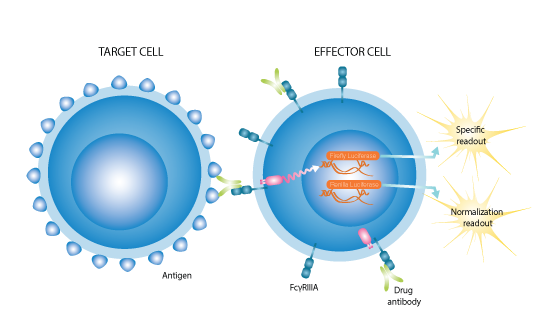
Further understanding of TNFα biology and its function in disease should contribute to a new generation of anti-TNFα antibodies with fewer side-effects. Being able to accurately measure any unwanted antibody-dependent cell-mediated cytotoxicity (ADCC) activity of such antibody-based drug candidates is essential in the development of new therapeutics. Svar offers a sensitive and specific cell-based iLite® ADCC mTNFα assay which is ideal for determining the potential of an antibody targeting membrane-bound TNFα to elicit ADCC.
The combination of cleverly engineered effector cells and homologous antigen-positive or antigen-negative target cells creates a system that offers unparalleled sensitivity for each target and is truly greater than the sum of its parts.