- Other Products
- CCP
Complement-dependent cytotoxicity (CDC) is an integral part of our immune defense system by activating the complement system to eliminate target cells. However, CDC dysregulation can contribute to autoimmune diseases and inflammatory pathologies.
In this blog post, we will explore the significance of CDC activity assessment in the development of therapeutics that modulate the complement system. By inhibiting or activating complement, these therapeutics can prevent unwanted complement activation in specific disease settings.
.jpg)
.jpg)
Complement-dependent cytotoxicity (CDC) is an integral part of our immune defense system by activating the complement system to eliminate target cells. However, CDC dysregulation can contribute to autoimmune diseases and inflammatory pathologies.
In this blog post, we will explore the significance of CDC activity assessment in the development of therapeutics that modulate the complement system. By inhibiting or activating complement, these therapeutics can prevent unwanted complement activation in specific disease settings.
The complement cascade plays a vital role in the immune response but can have harmful effects if dysregulated. Complement therapeutics offer a way to modulate this cascade, prevent terminal pathway formation, and mitigate cell damage. By targeting specific proteins, complement therapeutics effectively regulate the complement cascade's inflammatory and opsonic properties.
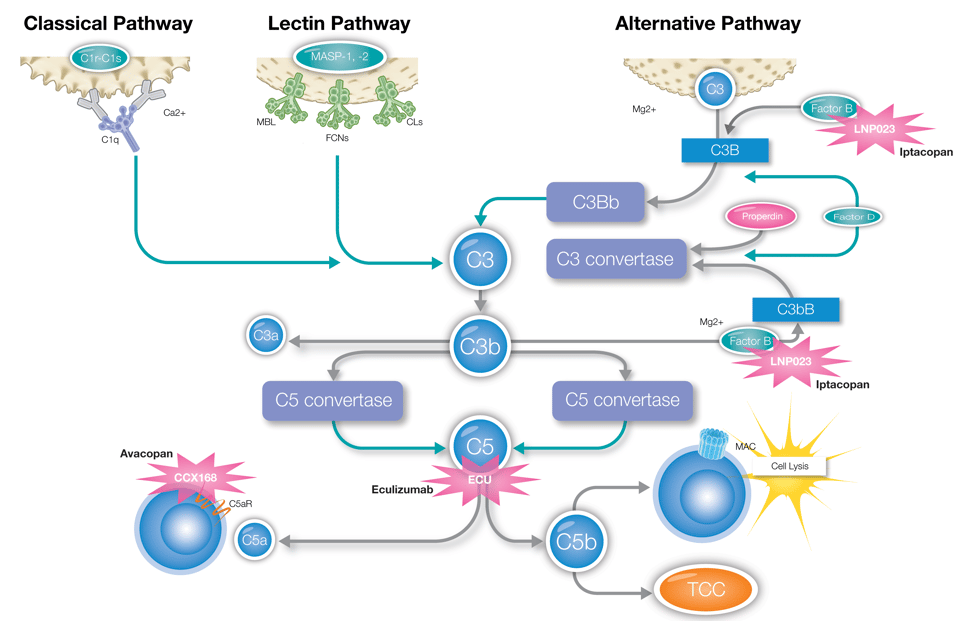
Assessing CDC activity during the development process offers several benefits. CDC is initiated by fixed C1q – the classical pathway's initiating component - and requires central components like C3 and C5 to occur. Inhibiting these components with complement-targeting therapeutics will impact CDC activity. Assessing CDC provides rapid and robust data on the complement-targeting therapeutic's ability to prevent cell lysis.
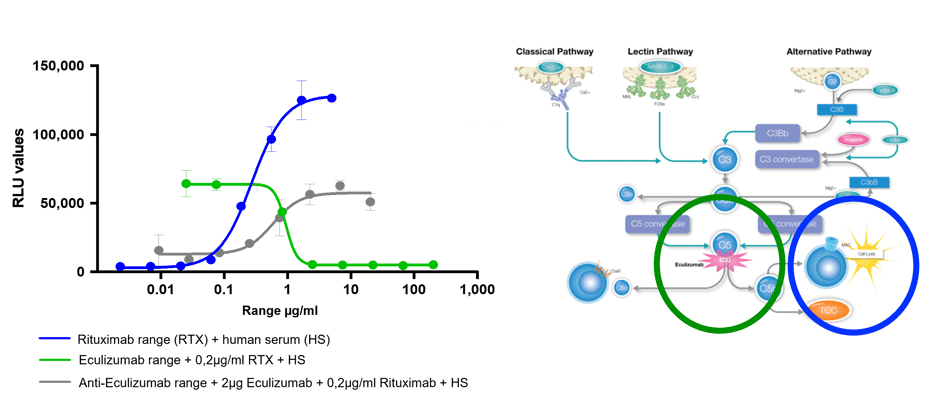
Bioassays are a great tool for assessing CDC. The newly developed iLite® CDC assay measures luciferase, released upon cell lysis, as an indicator of cytotoxicity. It provides high sensitivity and specificity, allowing for accurate quantification of CDC.
Additionally, the iLite CDC bioassay differentiates between complement-mediated and non-complement-mediated cell lysis since it requires the fixation of C1q. It is a simple and cost-effective approach that can be easily adapted to high-throughput screening. It doesn't require specialized facilities and trained personnel to handle radioactive materials. With its assay-ready format, you can obtain results within 5 hours.
Furthermore, the iLite CDC bioassay is highly customizable, meaning that our scientists can modify it to express a wide variety of surface antigens.
Understanding CDC and its role in complement therapeutics is vital for the development of effective treatments. By targeting the complement cascade and evaluating therapeutic activity through the innovative iLite CDC bioassay, scientists can ensure truly reflective Mechanism of Action (MoA) assessment and unlock new possibilities in the field of anti-complement therapies.