iLite AAV NAb Assessment
Reliable Detection of Neutralizing Antibodies against AAV Capsids
Reliable Detection of AAV Neutralizing Antibodies
AAV NAb Bioassays
Detection of neutralizing antibodies (NAbs) in AAV gene therapies is essential. Our AAV NAb cell-based reporter-gene assays, based on our proprietary iLite® technology, offer the tool you need for identifying and assessing Neutralizing Antibodies against AAV capsids.
iLite AAV NAb Reporter Gene Assays
Our AAV NAb assessment bioassays are designed to evaluate the immunogenicity of AAV capsids for different serotypes in gene therapy. They use a two-cell line setup: an AAV-responsive reporter cell and packaging cells for the vector serotype.
- Customization for any capsid – both traditional and recombinant
- Packaging cells eliminate the need for vector production
- Reduced incubation time for fast and high-quality results
- "Assay-ready" format for rapid and convenient use
- A secondary reporter gene that acts as an internal control, compensating for differences in cell number and matrix effects
Our AAV NAb Reporter-Gene Assays
iLite® AAV Responsive Reporter Assay Ready Cells
Reporter cell line designed to quantify AAV vector transduction efficiency and detect NAbs that impair gene delivery
iLite® AAV2 Packaging Assay Ready Cells
Engineered and optimized to produce AAV vectors of serotype 2 for use in NAb detection studies
iLite® AAV5 Packaging Assay Ready Cells
Engineered and optimized to produce AAV vectors of serotype 5 for use in NAb detection studies
iLite® AAV6 Packaging Assay Ready Cells
Engineered and optimized to produce AAV vectors of serotype 6 for use in NAb detection studies
iLite® AAV8 Packaging Assay Ready Cells
Engineered and optimized to produce AAV vectors of serotype 8 for use in NAb detection studies
iLite® AAV9 Packaging Assay Ready Cells
Engineered and optimized to produce AAV vectors of serotype 9 for use in NAb detection studies
Didn't find what you were looking for?
We offer products customized to your needs. Our custom iLite® reporter-gene assays can be adapted to almost any target. We also offer custom ELISA plates and other products. In addition, you can take advantage of our service offerings and let us do the work for you.
iLite News
iLite Downloads

Systematic Analysis of Fc Mutations Designed to Modulate Functional Activity
Read post
Therapeutic Antibodies Trimmed with GlycINATOR® Lose Antibody-Dependent Cell-mediated Cytotoxicity
Read post
Quantification of Bispecific Antibody Mediated T-cell Activation
Read post
IL-6 Biology, Inhibitors & COVID-19
Read post
Mechanism-linked Potency Assays Drive Gene Therapy Success
Read post
Addressing the Analytical Gaps in Modern Therapeutic Development
Read post-1.jpg?width=500)
Developing a Customizable Complement-Dependent Cytotoxicity Assay
Read post
Partnering for Success in a New Era of Advanced Therapeutics
Read post
The Immune Response to AAV Gene Therapy
Read post
How to Design Potency Assays for Gene Therapy Projects
Read post
A Cell-Based ADCC Bioassay Comparison
Read post
Quantification of the Neutralizing Antibody Response to Recombinant AAV Vectors
Read post
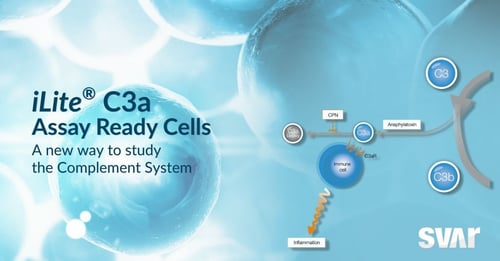
Svar Complement Assays: Analyzing How Drug Candidates Affect the Complement System
Read post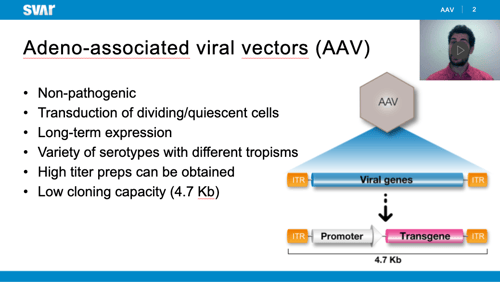
Assay Development in AAV Therapies: Engineering iLite® Cell Lines for Specific and Analytical Applic...
Read post
Measuring C5a With a Functional Bioassay
Read postContact our iLite team
Write us a message and a product specialist will get in touch